NSPKU England 2026 conference

This year the conference was held at the gorgeous Crewe Hotel and Spa, and I had a great time. Many thanks to all of those involved in creating a memorable conference.
There was so much chat and catching up that most of my notes were taken quickly. The following update is mostly photos taken on the day. I’ve organised them by talk and given comments needed. I hope you enjoy.
Future treatments update
Professor Anita MacDonald gave her usual update on the status of sapropterin use and possible new treatments for NSPKU.
As Anita said, “A minority of people have done well on Sapropterin (Kuvan), but we need other options.”
Much of this follows on her update from Wales six months ago, and I recommend reading that blog for full background.

There was still a way to go on registering other new treatments for PKU in the UK. This has been slowed by the UK’s exit from the EU.
Sepiapterin
Sepiapterin (Sephience) is similar to sapropterin, but has a greater success rate. See more here:

It has been approved for use in the EU, but the UK requires a separate licence and approval now. We understand the company will be applying for licence at uk end of 2026, approval may intake several years.
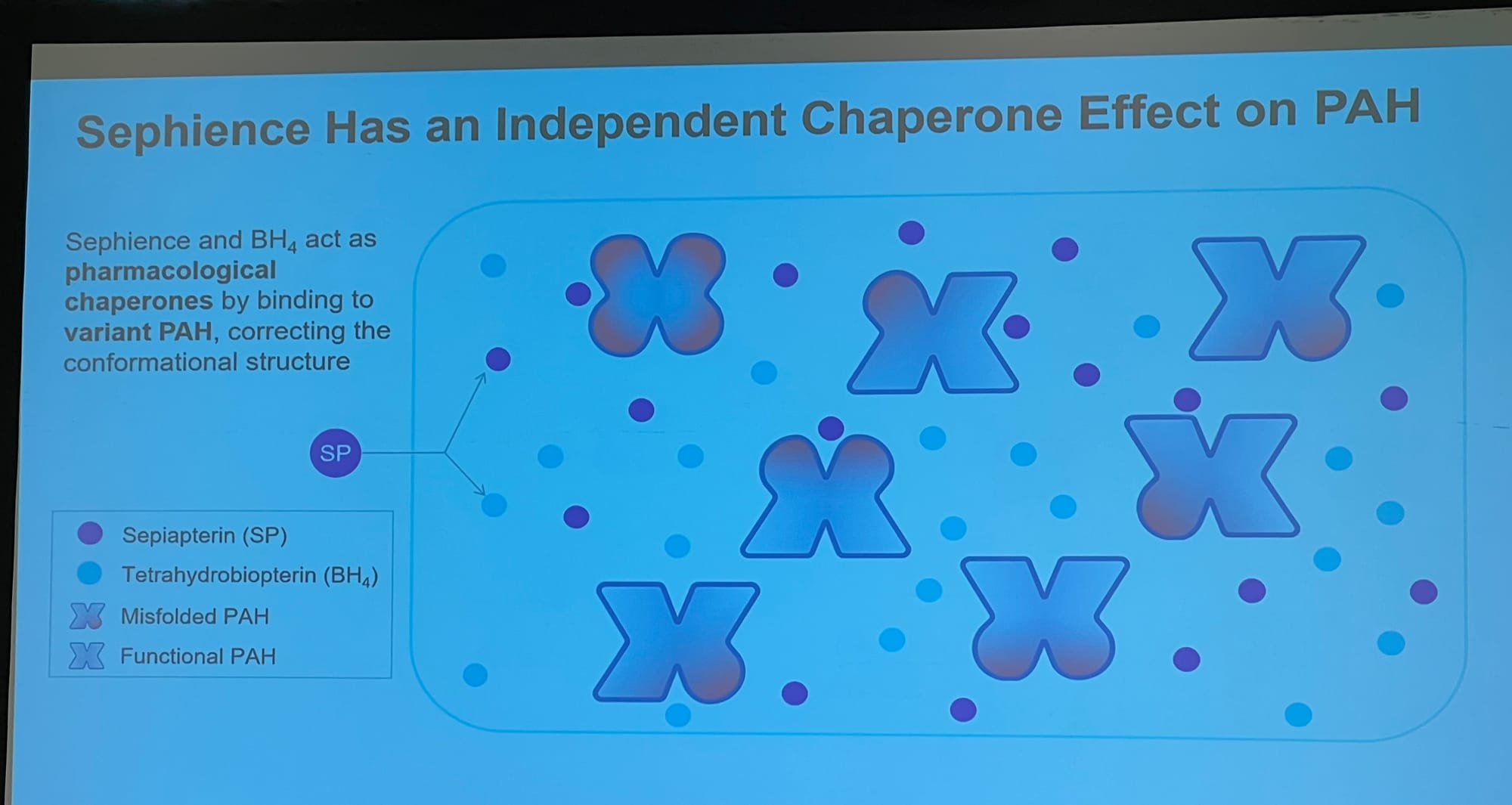
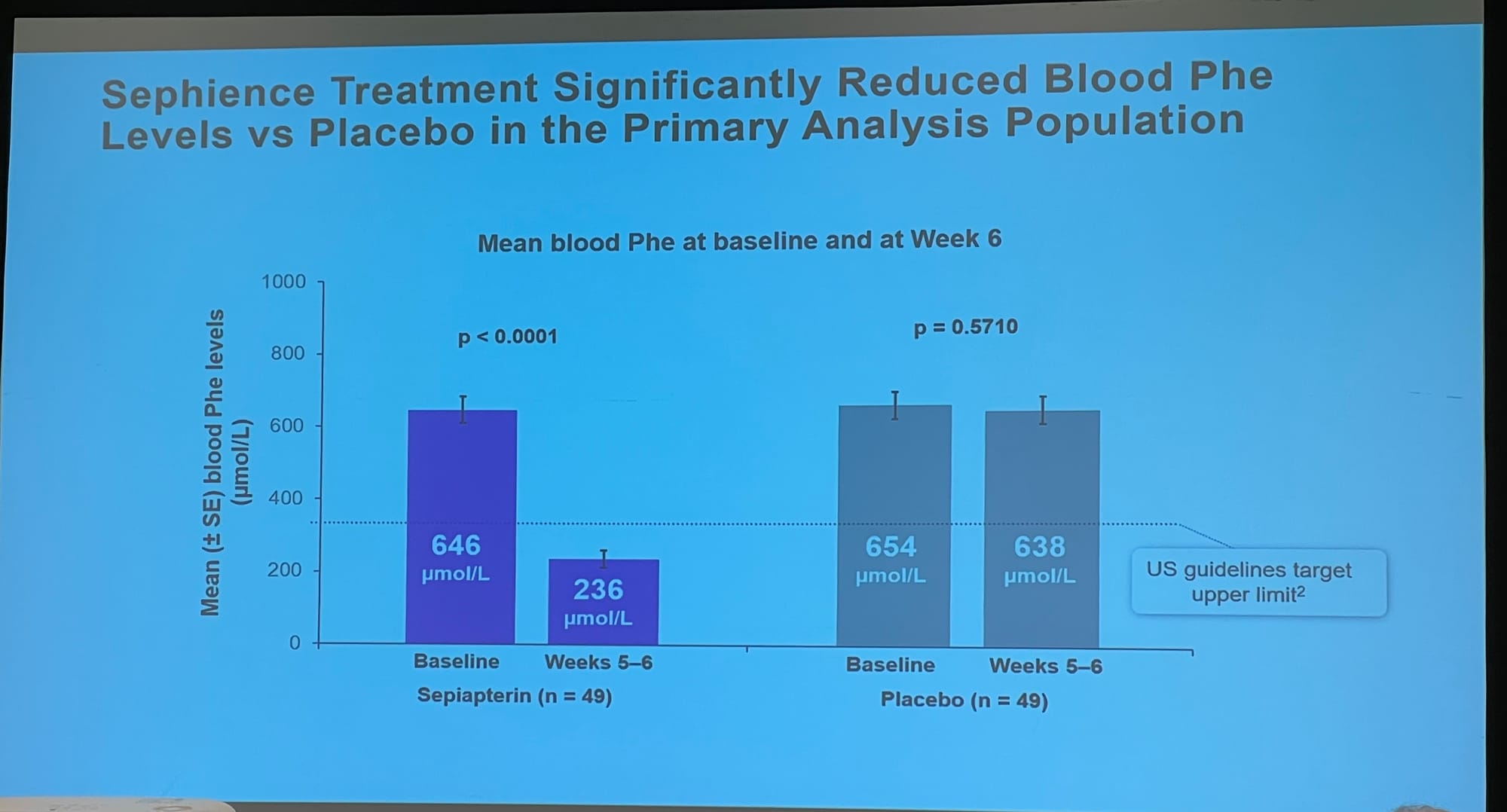
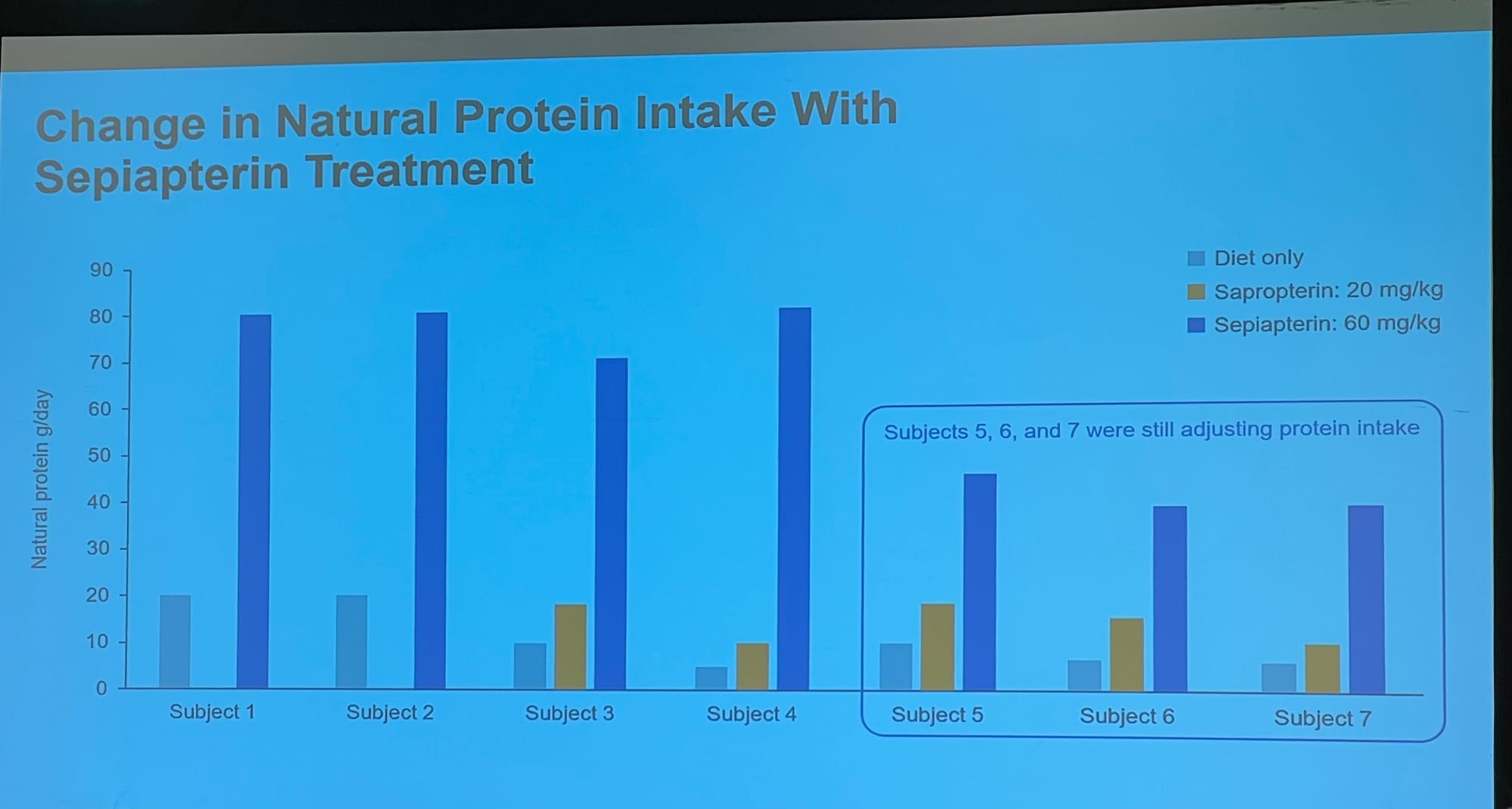

Slides showing: 1: how Sephience works by helping reshape our misshapen enzyme; 2: the reduction in blood phe in a recent trial; 3: the change in protein allowance; 4: Personal feedback on the effects of the treatment.
Kidney treatment options
Both options for possible kidney days treatments are still going through their trials.
JNT-517 (Repinatrabit)
Otsuka are putting JNT-517 through the final trials before going for a licence. Results still a year away at best.
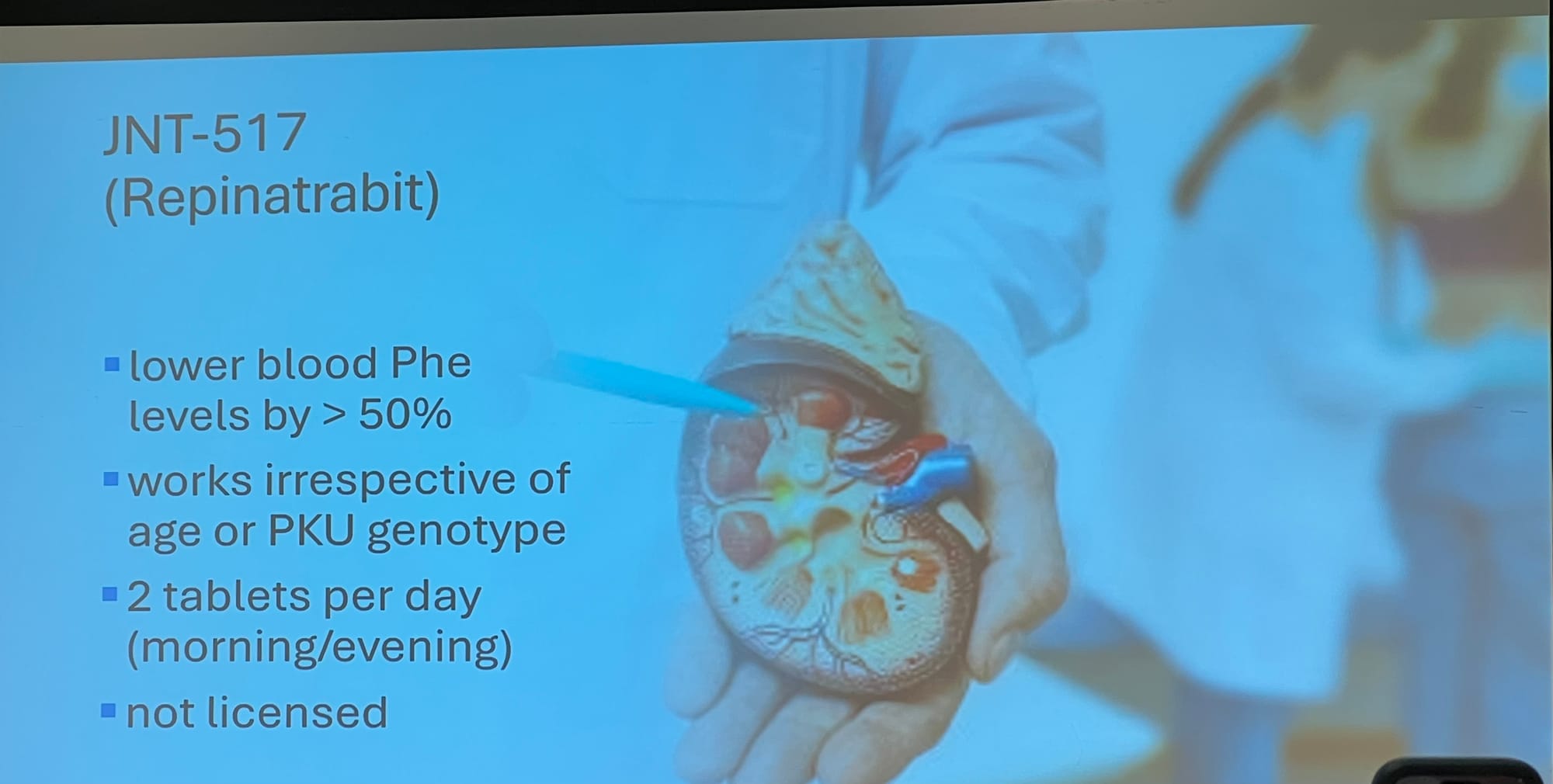
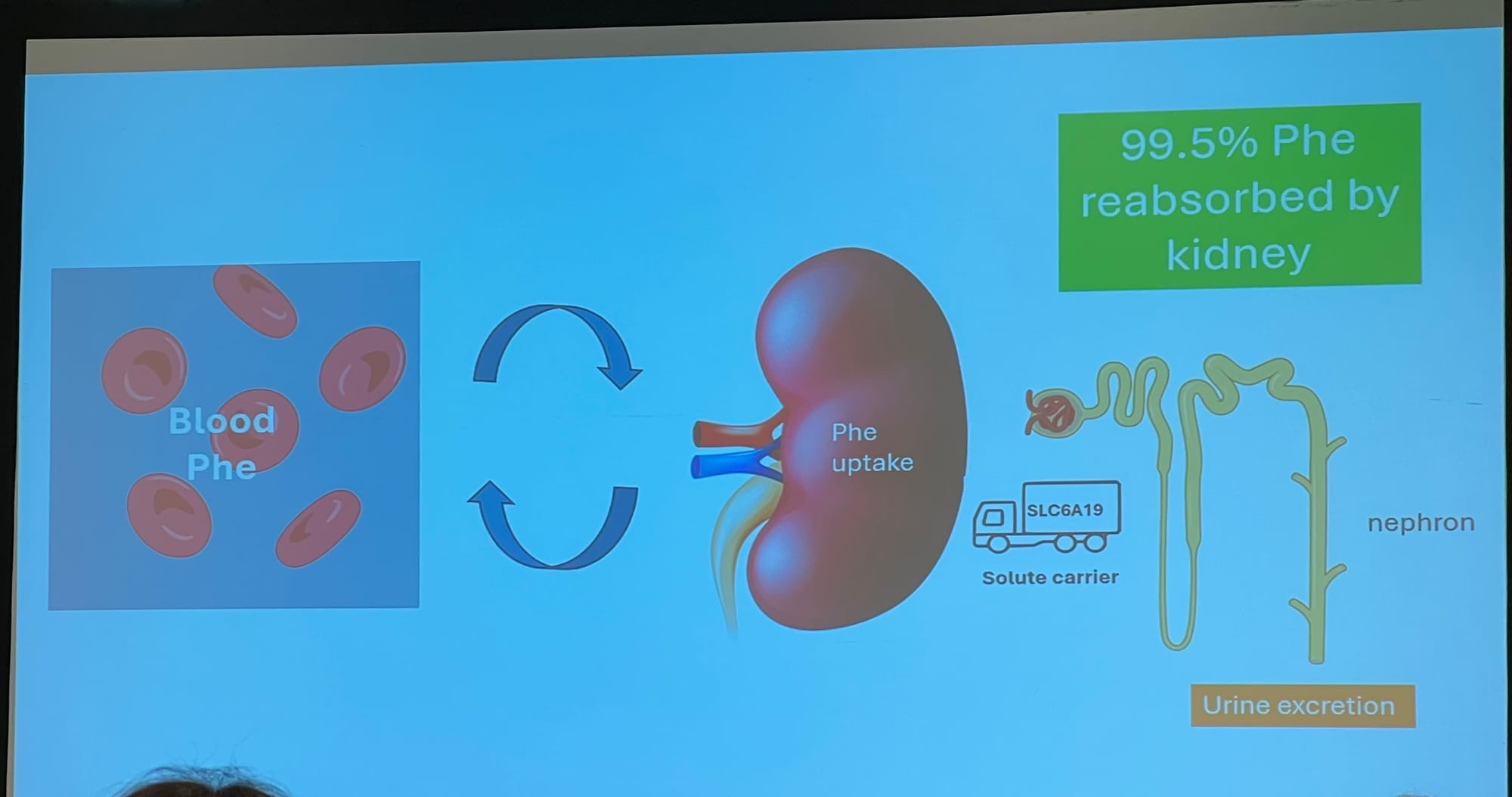
Slides detailing: 1: current knowledge on JNT-517, and 2: how JNT-517 works.
MZE782
Maze Therapeutics going to trial their kidney treatment option in UK soon. As this hasn’t started, we are unlikely to see anything about availability for several years.

Home Blood phe monitoring
There was an update by Dr Pinto on the trials of the Egoo machine. This is a device, referred to in the slides at POCT, which allows for a result at home in 30 minutes. For obvious reasons, it has long been desired by the PKU community.
Dr Pinto and team ran trials in homes to compare the real-world use of the device against traditional monitoring. At present, we take dried blood spots (DBS) which are posted to the clinics for analysis.


Slides showing: 1: Family feedback from the trail, and 2: Conclusions from using the Egoo (POCT) machine vs current testing (DBS).
These results are encouraging, but there is still a long way to go before the machine is approved in the UK, or by the NHS. Do note: the last time I checked (early 2026) this machine was not available to buy privately in the UK either.
Buy me a cuppa ☕ — your support helps keep this blog free, and helps to me write about PKU, brain injury & mental health.
Psychology update, Megan Staines
Megan Staines gave an update on her research into PKU and Neuropsychology.


Slide 1: Background & aims for the study, and 2: conclusions.
PKU, Exercise, & quality of life
Annie Skidmore gave a much anticipated update on her research into PKU and Exercise.
Stop Press: Annie is running the London Marathon this weekend! (26 April 2026)




Slides on Annie Skidmores work into PKU and Physical Activity & Exercise (PA/E).
Annie’s most startling finding so far was that many with PKU reported significant barriers to Physical activity and needed support.
However, most people in a position to provide that support did not see the barriers, and missed the opportunity to provide support.
PKU and the gut microbiome
Catarina Rodrigues gave a presentation on her planned research into PKU & poo—to the giggles of the young (and not so young) in the audience.
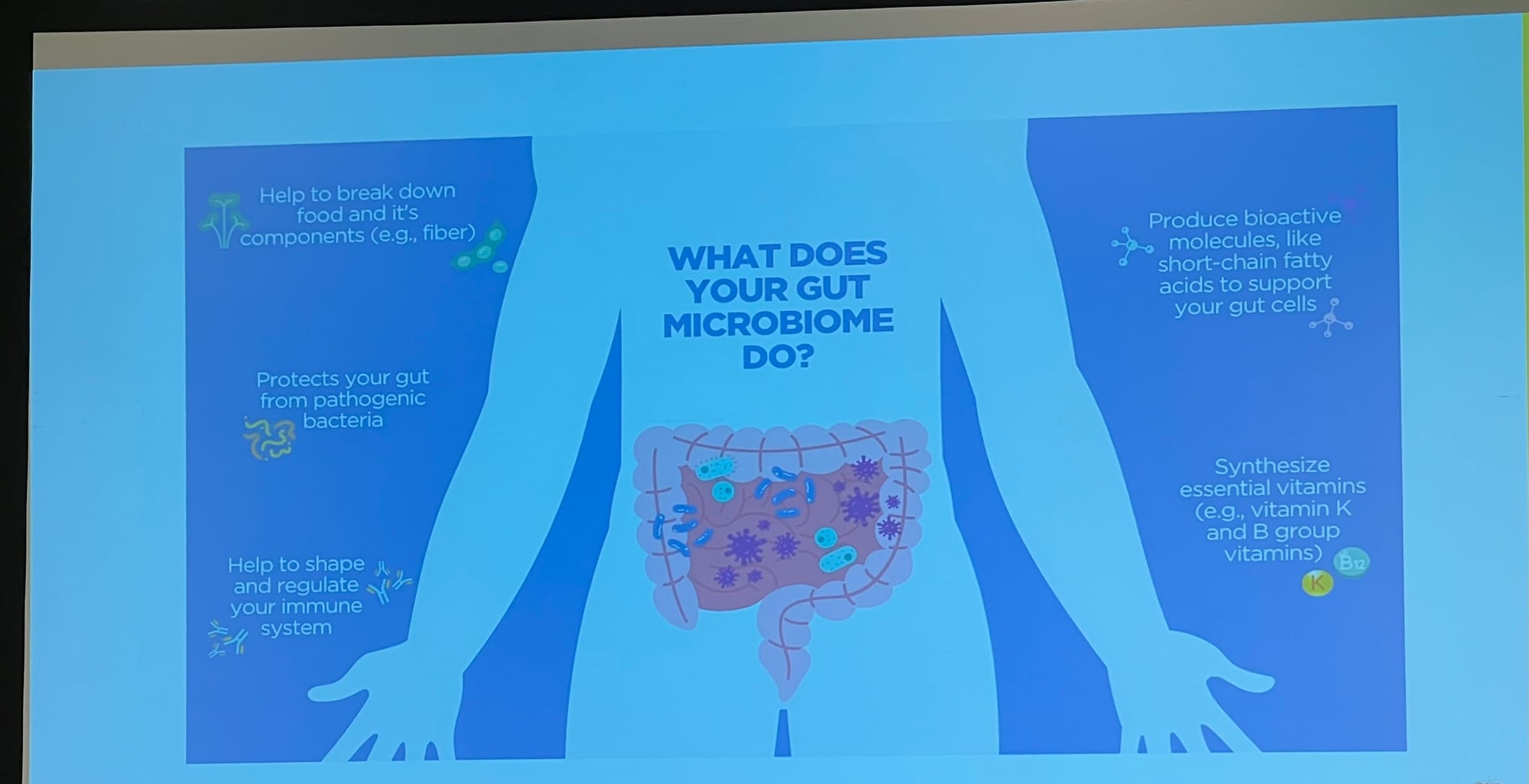

Slides 1: Detail on the importance of the gut microbiome, and 2: the scope of the study.
Please help with updates
This was such a busy conferece that I missed some sections. If you are willing to help fill in those gaps then please get in touch!


Member discussion